Medical Titanium Wire
Medical Titanium Wire is a high-purity titanium and titanium alloy wire specifically designed for implants and surgical fixation devices.
Unlike industrial titanium wire, medical grade titanium wire must meet four critical requirements: biocompatibility, low interstitial element (ELI) control, medical-clean finish, and regulatory compliance. These ensure long-term implant safety, prevent metal ion release, and guarantee compatibility with MRI/CT imaging.
As a professional Medical Grade Titanium Wire Manufacturer, Chalco supplies wires compliant with ASTM F67, ASTM F136, ASTM F2063, and ISO 13485 standards, offering customized specifications and fast delivery.Request a Sample

Why Choose Medical Grade Titanium Wire?
Not all titanium wires are suitable for medical devices. Medical Grade Titanium Wire differs fundamentally from industrial titanium wire in composition, cleanliness, and testing standards. Using the wrong material can compromise implant safety, cause regulatory non-compliance, and lead to registration failure or costly recalls.
Biocompatibility
Medical-grade wire passes ISO 10993 biological safety tests, ensuring no allergic reactions, toxicity, or tissue rejection after implantation.
Low Interstitial Element (ELI) Control
Strict control of O, N, and H content guarantees material toughness and reduces the risk of brittle fracture.
Medical-Clean Manufacturing & Sterilization Ready
Produced under vacuum annealing and ultrasonic cleaning, packaged in Class 10,000 cleanroom conditions, and ready for EO sterilization.
Regulatory Compliance & Full Traceability
Compliant with ASTM F67 / F136 / F2063 and ISO 13485, each batch is supplied with EN 10204 3.1 certificates and supports UDI batch traceability.
Don't risk delays or recalls by using the wrong material. Choose ASTM/ISO-certified Medical Grade Titanium Wire to ensure compliance, reduce costs, and guarantee patient safety.
Contact Chalco now to get the compliance checklist and a custom quotation!Contact Us
Industrial vs Medical Grade Titanium Wire: One Table to Understand the Difference
Before making a purchase decision, it is essential to understand the core differences between industrial titanium wire and medical grade titanium wire. The table below clearly compares standards, cleanliness, and regulatory requirements to help you make a compliant choice.


| Dimension | Industrial Titanium Wire | Medical Grade Titanium Wire |
| Applicable Standards | ASTM B348 / B863 — For general engineering; no implant clauses | ASTM F67 / F136 / F2063 & ISO 5832 — Designed specifically for implants |
| Interstitial Element (ELI) Control | Oxygen/Nitrogen/Hydrogen limits are loose → risk of embrittlement | Strict low ELI content, higher toughness, superior fatigue resistance |
| Biocompatibility | Typically not ISO 10993 tested; potential allergy/toxicity risk | Compliant with ISO 10993, MRI/CT safe with no imaging artifacts |
| Cleanliness & Packaging | Basic acid pickling; bulk coils | Vacuum annealing + ultrasonic cleaning + Class 10k cleanroom packaging |
| Quality System & Traceability | ISO 9001 or none | ISO 13485 certified + UDI batch traceability |
| Regulatory Risk | Hard to pass FDA/CE → delays, recalls | Passes audits in one go, shortens approval time |
If your products are intended for implantable or critical medical applications, medical grade titanium wire is the only compliant option. Do not risk regulatory delays or costly recalls—choose ASTM/ISO-certified medical titanium wire from Chalco for full compliance, patient safety, and faster delivery.Submit Specifications
Interested in industrial titanium wire or 3D printing titanium wire feedstock? Click here to explore detailed specifications.
Compliance Thresholds & Global Standards — The "Safety Line" for Medical Titanium Wire
To enter the global implantable device market, medical grade titanium wire must pass rigorous multi-level approvals, including ASTM, ISO, and FDA/CE. Below are the critical standards and regulatory checkpoints every supplier and buyer must understand.
1. ASTM & ISO Implant Standards
ASTM F67 — Commercially pure titanium (Grade 1–4), for sternal closure and orthopedic fixation
ASTM F136 — Ti-6Al-4V ELI alloy, ideal for bone cerclage, K-wires, and high-load implants
ASTM F2063 — Nitinol alloy, used in guidewires, orthodontic archwires, and vascular stents
ISO 5832-2 / -3 — Titanium and titanium alloy implant standards designated under EU MDR
2. Quality System & Traceability
ISO 13485 — Medical device QMS; Chalco audited by SGS
UDI Batch Traceability — Serialized tracking, compatible with FDA GUDID upload
EN 10204 3.1 Mill Test Certificate — Each batch ships with chemical and mechanical reports
3. Biocompatibility & Cleanliness
ISO 10993 Full Panel Testing — Hemolysis, cytotoxicity, skin irritation
Class 10,000 Cleanroom Packaging + EO sterilization-ready
→ Cuts terminal sterilization verification costs and time
Choosing medical titanium wire that complies with ASTM F136 / F67 / F2063 and ISO 13485 ensures a smooth regulatory path. You'll avoid redundant testing, reduce registration delays, and accelerate FDA 510(k) or CE MDR submissions.Request Report
Chalco Medical Titanium Wire Solutions: Simpler Compliance, Faster Delivery
Leveraging full industrial integration and an ISO 13485-certified quality system, Chalco offers a zero-risk, end-to-end solution for medical grade titanium wire — from R&D to mass production and global delivery.
Full Chain Control: From Melting to Packaging
- In-house Vacuum Arc Remelting (VAR) :Ensures low ELI (extra-low interstitials)
- Multi-stage cold drawing + vacuum annealing:Excellent fatigue resistance, precise diameter tolerance
- Class 10k cleanroom ultrasonic cleaning & dual sterile packaging:Ready for sterile production


Rapid Delivery & Flexible Customization
- Samples in 2 weeks, bulk delivery in 4–6 weeks; standard sizes in stock year-round
- Full size range 0.08–6.0 mm, customizable hardness: annealed / semi-hard / hard
- Dedicated engineering team:Feasibility report & quote in 48 hours
Stringent Testing & Full Traceability
- ICP-OES chemical analysis, fatigue life testing, surface roughness inspection
- Each batch ships with EN 10204 3.1 MTC + ISO 10993 biocompatibility report
- UDI barcode bound to ERP:Traceable to VAR heat batch in under 5 minutes


Your Global Compliance Accelerator
- Complete ASTM F67 / F136 / F2063 data packages + ISO 13485 certification
- Regulatory advisors assist with FDA 510(k) and EU MDR technical dossiers
- Proven success in 30+ countries across North America, EU, Japan, Korea, Middle East
With Chalco's one-stop solution, from raw titanium to certified documentation, you can reduce registration time by 30% and total procurement cost by 20%.Request Sample
Choose the Right Titanium Alloy for Your Medical Application
Different medical implants require different performance characteristics from titanium wire.
Chalco offers three main types of medical grade titanium wire to meet the needs of orthopedic fixation, sternal closure, vascular interventions, and orthodontics:
Grade 2 Pure Titanium Wire (ASTM F67)
Key Features: Excellent ductility and corrosion resistance
Applications: Sternal closure wire, orthopedic sutures, low-load fixation components
Standards: ASTM F67, ISO 5832-2


Ti-6Al-4V ELI Titanium Alloy Wire (ASTM F136)
Key Features: High strength with extra-low interstitials (ELI)
Applications: Cerclage cables, Kirschner wires (K-wires), intramedullary nails, orthopedic ligature wires
Standards: ASTM F136, ISO 5832-3
Nitinol Memory Alloy Wire (ASTM F2063)
Key Features: Shape memory and superelasticity; low rebound force
Applications: Guidewires for vascular intervention, orthodontic archwires, cardiac stent frameworks
Standards: ASTM F2063

Typical Applications of Medical Titanium Wire
Chalco's medical grade titanium wires are trusted by medical device manufacturers worldwide. Below are four typical application scenarios, demonstrating how we solve pain points and enhance device performance through alloy selection and clean manufacturing.
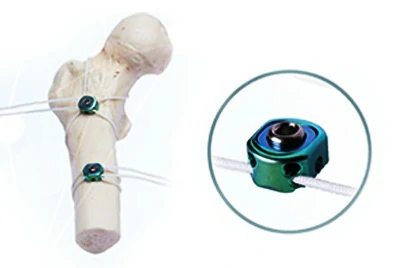
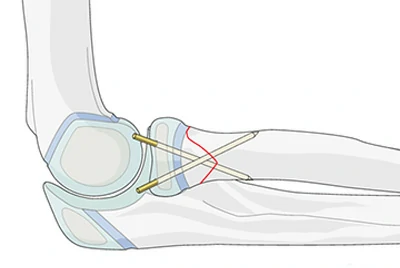
Orthopedic Fixation – Bone Cerclage & K-Wires
Bone cerclage systems are subjected to long-term multiaxial cyclic loads, and standard industrial wire often fails due to fatigue.
Chalco uses Ti-6Al-4V ELI (ASTM F136) with vacuum annealing, improving fatigue life by 35% while maintaining low interstitial levels (ELI) for regulatory compliance.
Case study: A German orthopedic company achieved CE MDR approval in a single submission, accelerating product launch by 4 months.
[See Bone Cerclage Wire Details]
Sternal Closure Wire
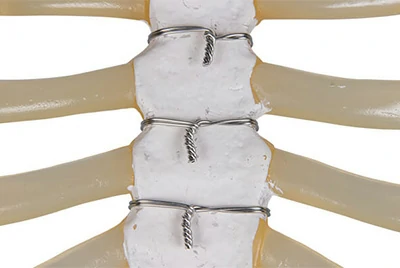
Post-cardiac surgery requires soft, MRI-compatible closure materials.
Chalco provides Grade 2 pure titanium wire (ASTM F67) with Ra ≤ 0.6 µm medical-clean surface, minimizing tissue irritation and ensuring artifact-free imaging.
Case study: A U.S. cardiovascular device brand reported a 20% drop in post-op complications, adopted by 5 leading hospitals.

Vascular Intervention Guidewire
Guidewires require both superelasticity and pre-formability.
Chalco supplies Nitinol wire (ASTM F2063) as thin as 0.127 mm, with customizable A_f transformation temperatures and ≥10⁷ bending cycles.
Case study: An Asian OEM passed FDA 510(k) approval with improved yield by 18%.
[See Nitinol Wire Details]


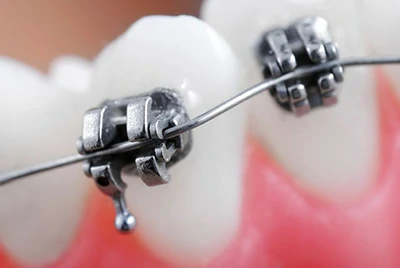
Dental Orthodontic Archwire
Orthodontic wires must maintain long-term shape with low friction.
Chalco offers β-Titanium and Nitinol hybrid wires, with TiN coating that reduces friction by 25%.
Case study: A European orthodontic brand reduced patient revisit rates by 15% and increased market share.
[See Orthodontic Wire Details]
Still planning your next device? Send us your design and technical requirements, and we'll provide alloy recommendations and sampling solutions within 24 hours.Submit Specifications
Quality System & Manufacturing Capability
From melting to packaging, Chalco operates under ISO 13485, implementing full-process testing and batch traceability to ensure every meter of medical grade titanium wire meets the highest regulatory standards.
Integrated Manufacturing
Vacuum Arc Remelting (VAR) – Ensures low interstitial elements (ELI) for implant-grade purity
Multi-pass precision drawing + vacuum annealing – Diameter tolerance ±0.02 mm
Class 10,000 cleanroom ultrasonic cleaning + double-layer sterile packaging
Annual capacity of 800 tons of medical titanium wire, covering 0.08 mm to 6.0 mm full diameter range
Rigorous Testing Suite
ICP-OES elemental analysis – Traceability for 15 elements
Fatigue life testing – ≥ 10⁷ cycles for Ti-6Al-4V ELI wires
AOI surface inspection – Ra ≤ 0.8 µm, free from scratches or defects
ISO 10993 biocompatibility reports + SGS / BV third-party certifications
Traceability & Documentation
UDI barcoding linked to ERP – Trace each spool to its VAR batch in 5 seconds
EN 10204 3.1 Mill Test Certificate (MTC) included with every shipment
Full compliance documentation: ASTM / ISO test reports, EO/γ sterilization validation files
Strict regulations? Tight lead times?
Let Chalco's ISO 13485-certified factory be your regulatory fortress—performance, paperwork, and traceability—all in one.Request Full Report
FAQs
Can industrial titanium wire be used for implants?
No. Industrial titanium wire does not meet the low interstitial requirements of ASTM F67 / F136 or pass ISO 10993 biocompatibility tests. Using it in implants may lead to regulatory rejection and metal ion release risks. Always choose medical grade titanium wire compliant with implant standards.
What's the minimum order quantity (MOQ) and lead time?
Standard MOQ is 5 kg. Sample delivery in 2 weeks, bulk orders shipped within 4–6 weeks. In-stock items can be dispatched within 3 days.
What certifications and test reports are provided?
Each batch includes:
- EN 10204 3.1 Mill Test Certificate (MTC)
- ISO 10993 biocompatibility reports
- ASTM / ISO mechanical property test results
- Optional: SGS / BV third-party verification
Can you customize diameters, tempers, or ultra-fine wires?
Yes. We offer full customization from 0.08 mm to 6.0 mm in Soft, Semi-Hard, or Hard conditions. Micro-diameter Nitinol as small as 0.05 mm is also available for bulk supply.
Do you offer sterilization-ready packaging?
Yes. Cleanroom Class 10k packaging includes double-layer PE bags + aluminum foil vacuum sealing, suitable for EO (Ethylene Oxide) sterilization.
Still have questions? Talk to our engineers 1-on-1.Contact Now




